Reagent Packaging for Diagnostic Applications
Validated reagent packaging for IVD and POC diagnostic consumables. Controlled-environment dispensing, blister and pouch packaging, and precision fill accuracy from pilot to commercial scale.


Life Science Outsourcing provides precision reagent packaging services for IVD, POC, and clinical diagnostic manufacturers — ensuring product integrity, fill accuracy, and regulatory compliance from pilot scale through commercial production.

Precision Reagent Packaging: Protecting Integrity, Ensuring Accuracy
In the world of diagnostics, precision isn't optional — it's essential. The right reagent packaging solution protects sensitive chemistries, maintains fill accuracy at volume, and ensures your consumable performs consistently from the first unit off the line to the ten-millionth.
LSO's reagent packaging capabilities were purpose-built for diagnostics through the J-Pac Medical acquisition in January 2023. Our controlled-environment manufacturing, precision dispensing systems, and diagnostics-specific DFM expertise mean your reagents are handled by a team that understands what's at stake when fill volumes drift or seal integrity fails.

What Is Precision Reagent Packaging?
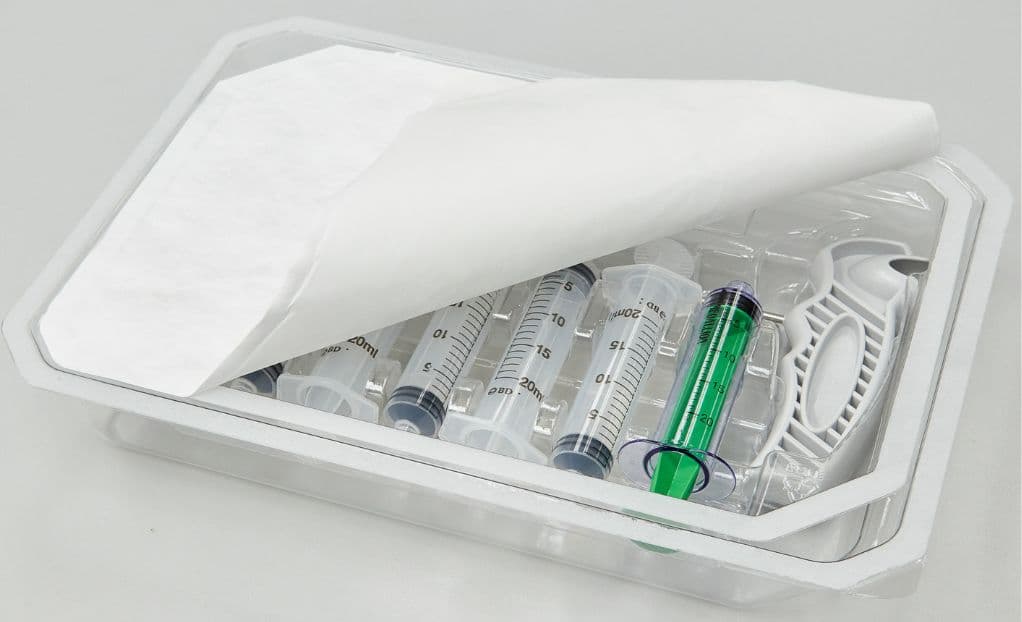
Precision reagent packaging ensures secure containment, stability, and compliance for diagnostic reagents across multiple formats — blister packs, pouches, vials, and custom configurations. Key capabilities include:
Controlled-environment dispensing with validated fill accuracy for your specific reagent chemistry. Blister packaging for liquid and lyophilized reagents with controlled fill volumes and seal integrity. Pouch and tray packaging for diagnostic kits and consumable sets. Nitrogen blanketing and moisture barrier options for stability-sensitive formulations. Complete lot traceability and documentation for regulatory submission.

Who Needs Precision Reagent Packaging?
Reagent packaging services are used by IVD and POC diagnostic manufacturers who need validated, controlled-environment packaging for their reagent consumables. Typical customers include:
IVD manufacturers scaling from clinical trials to commercial production. POC test kit companies transitioning from R&D prototyping to validated manufacturing. Reagent suppliers who need blister or pouch packaging with precision fill accuracy. Diagnostic kit assemblers requiring complete kit assembly with reagent integration. Companies whose current packaging partner can't maintain fill accuracy at volume.
How Does Precision Reagent Packaging Work?
LSO's reagent packaging process follows a structured path from technical assessment through commercial production:
1. Technical Assessment — Review your reagent chemistry, fill volume requirements, packaging format, and stability constraints. 2. Process Development — Develop dispensing parameters, seal specifications, and environmental controls specific to your reagent. 3. Validation — IQ/OQ/PQ for dispensing equipment, sealing processes, and environmental conditions. 4. Pilot Production — Initial runs with full QC to verify fill accuracy, seal integrity, and yield rates. 5. Commercial Production — Ongoing production with real-time quality monitoring and scheduled shipments.
Standards and Quality Controls
Reagent packaging at LSO is performed under our FDA-registered, ISO 13485 certified quality system. Applicable standards include ISO 11607 for sterile barrier packaging, FDA 21 CFR Part 820 Quality System Regulation, and specific ASTM test methods for seal strength and package integrity. All processes are validated and documented to support your regulatory submission — whether FDA 510(k), De Novo, or international filings.
Why LSO for Reagent Packaging?
Most contract manufacturers treat reagent packaging as an afterthought — an extension of their general medical device packaging line. LSO's diagnostics capabilities were purpose-built through the J-Pac acquisition, not adapted from general manufacturing. Our team understands that a packaging defect on a diagnostic consumable isn't just a cosmetic issue — it's a failed test result.
Beyond packaging, LSO provides integrated services that eliminate vendor handoffs: cartridge assembly, sterilization management, labeling and serialization, and fulfillment — all under one quality system. Your reagent goes from filling to finished goods without leaving our controlled environment.
FDA-Registered
Manufacturing Facility
ISO 13485
Certified Quality System
12 Cleanrooms
Class 5–8
125,000+ Sq Ft
Regulated Manufacturing
3 Facilities
CA | NH | Costa Rica
Ready to get started?
Talk to a specialist about reagent packaging for diagnostic applications.
